Of course, you can also reach us via e-mail or our contact form. We will get in touch with you as soon as possible.
Identifying potential defects as early as the product development process and preventing them from occurring in the first place – what sounds like a dream becomes reality with the Failure Mode and Effects Analysis (FMEA). Read here what exactly an FMEA is, how it is carried out and which types can be differentiated between:
Failure Mode and Effects Analysis (FMEA) is an analytical method in quality management to find possible product defects before they occur. It is also known as "Impact Analysis".
Appropriate measures can be taken within the framework of the FMEA to prevent identified potential defects in advance. In this process, the product or process to be evaluated is assessed in terms of its impact on the customer, the probability of occurrence and the probability of detection using key figures. In this way, the process can be dealt with appropriately using suitable methods.
The goal of failure mode and effect analysis is to avoid errors instead of discovering them after the fact and then having to correct them, which may be costly. It also identifies potential weak points and critical components. This helps to avoid consequential costs in production or at the end user. In addition, the knowledge gained from the FMEA can be reused in other new products.
FMEA offers further advantages in the assessment of risks, through a shortened development time, lowering of costs as well as the effort for faults, and finally in the achievement of the company's political quality objectives.
The FMEA is divided into different areas depending on the time of application and the affected object:
The construction or design FMEA (construction failure mode and effects analysis) is a subarea of the FMEA that examines a specific product. It is used as part of the development, design and planning of new products and therefore takes place in particular in the early product planning phase.
The aim of the design fmea is to develop measures that contribute to reliability, safety and availability. An interdisciplinary team with participants from all departments involved carries out the FMEA. Focal points of the analysis are: Reliability, functionality, geometry, material selection, manufacturability, testability and serviceability.
The process FMEA is a subarea of the FMEA that deals with the investigation of a specific process. Triggers of a process FMEA can be, for example, new designs, technologies or processes, the change of an existing process or also the application of a process in a new area. In particular, it relates to the areas of manufacturing, assembly and testing. Process FMEA is performed as part of the production planning phase and aims to create a smooth process.
For this purpose, all factors that could prevent or complicate such a flawless process flow are identified. In the process FMEA, the entire chain of action with all influences is considered. The manufacturing process with the factors suitability and safety is taken into consideration, as well as quality capability and process stability, and the determination of process control characteristics.
The system FMEA (also: system analysis) is intended to examine a system as a whole and check whether all the individual components in it interact in a functionally correct manner. Measures can be derived from the analysis, the implementation of which is intended to increase system safety, reliability, and availability.
So-called fault sequences describe how the system behaves when a fault condition occurs. This requires initial information that can be taken, for example, from a requirements specification or from quality planning via quality function deployment.


An interdisciplinary team with participants mainly from the design, development, production planning, production execution and quality management departments is responsible for the FMEA. Forms or appropriate software are required for the analysis. This ensures a clear and orderly presentation.
First, a possible failure location is found by applying past experience and the specific expertise of those involved. Then it is determined what type of defect it is, the defect is described and evaluated; here the task priority (AP) in the harmonized FMEA (according to AIAG and VDA) replaces the previous risk priority number (RPN). Subsequently, the cause of the failure should be identified. Once the cause of the defect has been identified, measures can usually already be taken to avoid the defect. The proposed solutions should focus on the avoidance of errors, and not merely on their detection.
Since the harmonization of the VDA and AIAG FMEA manuals, prioritization in FMEA has been based on a logic of task priorities. The concept includes a logically structured explanation for each task priority: Thus, tasks can be prioritized based on individual evaluations of the individual values of B, A, and E as well as their combinations. The advantage over the replaced risk priority number is that a need for action can thus be determined much more precisely and also more reliably.
The FMEA serves to increase technical reliability. In quality management, it must therefore be carried out before the product is produced - i.e. in the early development phase, in the so-called product development process. This is because preventive measures are most effective the earlier they are started. FMEA is used as a method especially in the aerospace, automotive and other industries. But FMEA has also found its way into quality assurance in project management and the service industry.
The quality management standard ISO 9001:2015 calls for more risk awareness. "Risk" is described here as the effect of uncertainty on an event. According to this standard, companies are required to identify and evaluate potential risks. FMEA as a common method for risk management can thus help to meet the requirements of ISO 9001:2015.
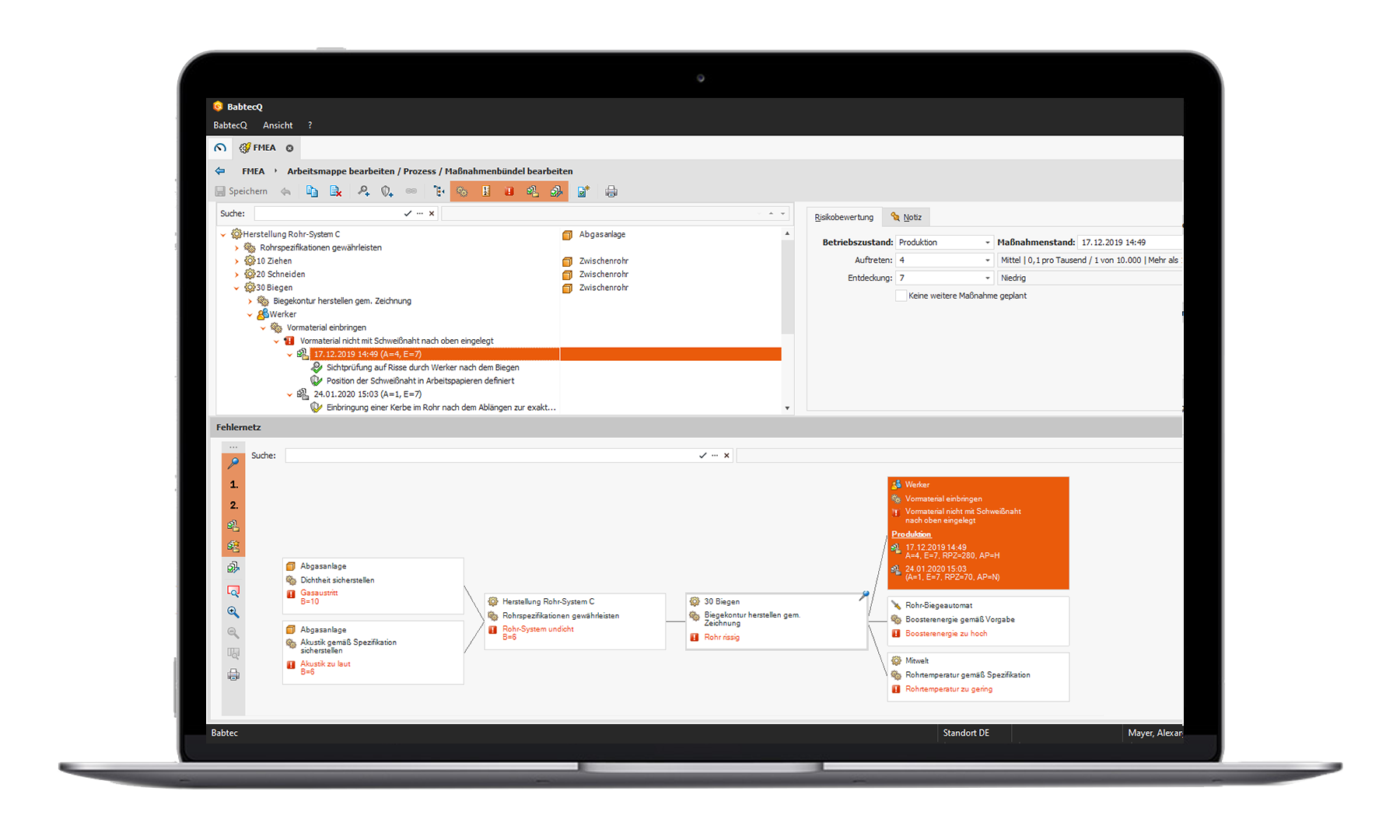
Our integrated solution supports you in performing and documenting standard-compliant design and process FMEAs. On the one hand, these can be systematically created and documented. On the other hand, our QM software BabtecQ also gives you the opportunity to use the findings efficiently for your further planning.
Knowing and systematically expanding the qualifications of one’s own employees is not only necessary, but also very sensible. This is the only way to ensure sustainable quality in the company and the satisfaction of the workforce. A helpful tool for this is the qualification matrix. You can read here... continue reading
Not everything you need to manufacture your own product you actually produce yourself. Instead, products from suppliers are used. The challenge here is that your own quality depends to a large extent on the quality of the supplier products and the reliability of the suppliers. So, which is the best?... continue reading
Anyone involved in quality management and QM systems has probably already come across the term skip lot procedure. This article explains what this sampling procedure is all about and why it can be useful. The term skip-lot (skip a lot) actually brings us pretty close to the general idea: Because this... continue reading
No Comments